
Ying (Maggie) Chen, MD, PhD, FASN
Associate Professor of Medicine (with Tenure); Associate Professor of Cell Biology & Physiology (with Tenure)
- Phone: 314-362-7603
- Fax: 314-747-5213
- Email: ychen32@nospam.wustl.edu
Additional Titles
- Director, Autosomal Dominant Tubulointerstitial Kidney Disease (ADTKD) Center
- Director, Nephrotic Syndrome Clinic (Nephcure Specialist)
Related Links
Location
Patients seen at
Center for Advanced Medicine
- Physical Address: 4921 Parkview Place, Saint Louis, MO, United States
-
Mailing Address:
4921 Parkview Place
Suite C, 5th Floor
St. Louis, MO 63110 - Phone: 314-362-7603
- Fax: 314-747-2460
Mailing Address
Division of Nephrology
-
Mailing Address:
660 S. Euclid Ave., CB 8126
St. Louis, MO 63110
In the news:
- R56 NIH Grant Awarded to Physician-Scientist Ying Maggie Chen
- Physician-Scientist Ying Maggie Chen Receives VA Merit Award
- Ying Maggie Chen Receives Grant from WashU Center of Regenerative Medicine
- Ying Maggie Chen Lab and Collaborators Uncover Biotherapeutic Properties of MANF Protein
- Collaborators Ying Maggie Chen and Srikanth Singamaneni Receive NIDDK R21 Grant to Develop Bio-needle Technology
- Nephrologist Ying Maggie Chen and Bio-Engineer Srikanth Singamaneni Receive OVCR Grant to Develop Bio-needle Technology to Detect AKI Biomarkers
- Physician-Scientist Ying Maggie Chen Elected to American Society for Clinical Investigation
Education
- Fellowship, Nephrology: Washington University School of Medicine, St. Louis, MO
- Residency, Internal Medicine: Saint Louis University School of Medicine, St. Louis, MO
- PhD Cell and Developmental Biology: Vanderbilt University School of Medicine, Nashville, TN
- Doctor of Medicine: Shanghai Medical University Shanghai, China Friedrich-Alexander-Universität Erlangen-Nürnberg School of Medicine Erlangen, Germany
Board Certifications
- American Board of Nephrology, 2010
- American Board of Internal Medicine, 2008
Recognition
Honors and awards
2021-2025
American Society Nephrology Grant Review Committee
Chartered member of VA Merit Award Review Committee
2025
NIH/NIDDK PBKD study section Ad Hoc reviewer
ASN Kidney Week Abstract Reviewer: Glomerular Diseases: Mechanisms, including Podocyte Biology
2024
VA Merit Award
R56, NIH/NIDDK
Rare Kidney Disease Clinical Advisory Council
ASN Kidney Week Abstract Reviewer: Glomerular Diseases: Mechanisms, including Podocyte Biology
George M. O’Brien Kidney Resource Alliance Opportunity Pool Program Reviewer
2023
Investigator Matching Grant, Washington University Center for Drug Discovery
ASN Kidney Week Abstract Reviewer: CKD (Non-Dialysis): Mechanisms
2022
Elected to the American Society of Clinical Investigation (ASCI)
R21, NIH/NIDDK
Washington University Office of the Vice Chancellor for Research Pilot grant
2021
US Patent: US 11,129,871
George M. O’Brien Kidney Core Center, P&F grant, NIH/NIDDK
2020
The Federal Advisory Committee (FAC) member
Fellow of the American Society of Nephrology (FASN)
George M. O’Brien Kidney Core Center, P&F grant, NIH/NIDDK
2018-2020
NIH/NIDDK R13 Grant Review Committee
2019
Investigator-Initiated Research Award, Department of Defense
Mallinckrodt Pharmaceuticals Challenge Grant
Investigator Matching Grant, Washington University Center for Drug Discovery
2018
Nephrotic Syndrome Specialist, nominated by NephCure Kidney International
US Patent: US 10,156,564
2017
R01, NIH/NIDDK
Children’s Discovery Institute hPSC Core Pilot Grant
2016
Renal Translational Innovation Grant, Washington University Division of Nephrology
2015
Clinical Scientist Development Award, Doris Duke Charitable Foundation
Early Career Development Award, Central Society for Clinical and Translational Research (CSCTR)
K Award, CSCTR and MWAFMR
Career Development Award, Nephrotic Syndrome Study Network (NEPTUNE)
R03 grant, NIH/NIDDK
2011
P30 (Pilot and Feasibility Grant), NIH/NIDDK
K Award, Combined Annual Meeting of CSCR (Central Society for Clinical Research) and MWAFMR (Midwestern Section of the American Federation for Medical Research)
2013
Faculty Scholar Award, Children’s Discovery Institute of Washington University and St. Louis Children’s Hospital
The Halpin Foundation-American Society of Nephrology for Kidney Research Grant
2010
K08 Clinical Scientist Development Award, NIH/NIDDK
2007
NIH Ruth L. Kirschstein National Research Service Award (NRSA, T32)
2003
Dissertation Enhancement Grant, Vanderbilt University School of Medicine
Clinical Interests
Primary nephrotic syndromes including focal segmental glomerulosclerosis, membranous nephropathy and minimal change disease; Alport Disease; Autosomal dominant tubulointerstitial kidney disease; Acid-base and electrolyte disorders.
Research Interests
Proteinuria; Podocytes; Endoplasmic Reticulum Stress; Organelle Biology; Renal Fibrosis
Dr. Chen’s research focus has been to develop novel therapeutics that target organelles, including endoplasmic reticulum (ER), mitochondria and lysosome to treat kidney disease. Her lab has pioneered discovery of urinary ER stress biomarkers. More importantly, her group has discovered podocyte ER calcium stabilizers as a new class of drugs to treat nephrotic syndrome, and MANF as a novel biotherapeutic protein to treat ADTKD. These research findings have been patented.
Dr. Chen’s research has been supported by NIH R01s, VA Merit Award and Department of Defense (DoD), as well as numerous Career Development Awards. She has been a chartered member of American Society of Nephrology and VA Merit Award grant review committees, and an ad hoc reviewer for NIDDK PBKD study section. She is an elected member of American Society of Clinical Investigation (ASCI).
Read more at the Y. Maggie Chen Lab website.
Publications
Li C, Fang YL, Chen Y. Beyond redox regulation: novel roles of TXNIP in the pathogenesis and therapeutic targeting of kidney disease. Am J Pathol doi.org/10.1016/j.ajpath.2024.12.011, 2024 PMID: 39814099
Kim Y*, Li C*, Gu CJ, Fang YL, Tycksen E, Puri A, Pietka TA, Sivapackiam J, Kidd K, Park SJ, Johnson BG, Kmoch S, Duffield JS, Bleyer AJ, Jackrel ME, Urano F, Sharma V, Lindahl M, Chen Y. MANF stimulates autophagy and restores mitochondrial homeostasis to treat autosomal dominant tubulointerstitial kidney disease in mice. Nat Commun 2023
Li C, Krothapalli S, Chen Y. Targeting endoplasmic reticulum for novel therapeutics and monitoring in acute kidney injury. Nephron 2023
Park SJ, Kim Y, Li C, Suh J, Sivapackiam J, Goncalves TM, Jarad G, Zhao G, Urano F, Sharma V, Chen Y. Blocking CHOP-dependent TXNIP shuttling to mitochondria attenuates albuminuria and mitigates kidney injury in nephrotic syndrome. Proc Natl Acad Sci 2022
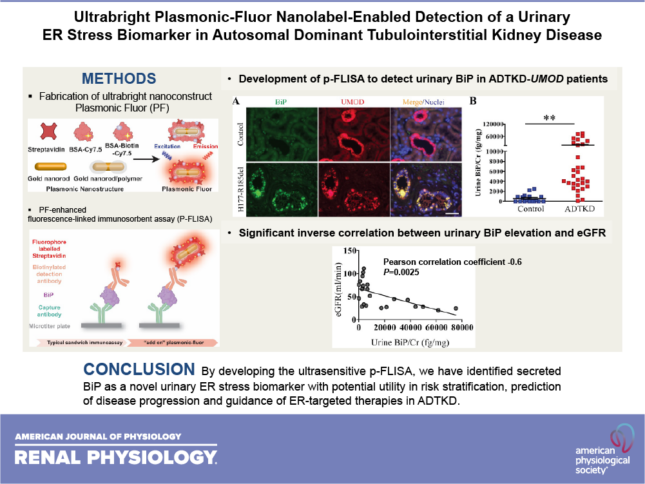
Kim Y, Wang ZY, Li C, Kidd K, Wang YX, Johnson B, Kmoch S, Morrissey J, Bleyer A, Duffield JS, Singamaneni S, Chen Y. Ultrabright plasmonic-fluor nanolabel-enabled detection of a urinary ER stress biomarker in autosomal dominant tubulointerstitial kidney disease. Am J Physiol Renal Physiol 321(2):F236-F244, 2021
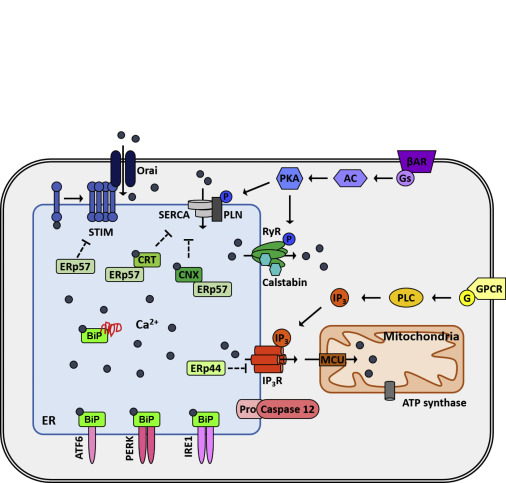
Park SJ, Li C, Chen Y. . Am J Pathol 191(2):256-265, 2021 PMCID: PMC7863132
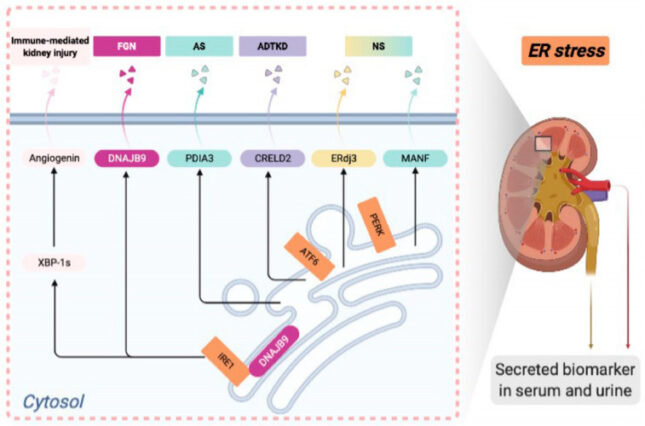
Li C, Chen Y. Endoplasmic reticulum-associated biomarkers for molecular phenotyping of rare kidney disease. Int J Mol Sci 22(4), 2021 PMCID: PMC7926397
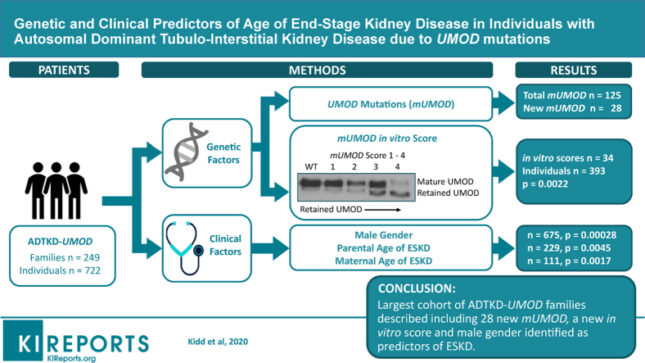
Kidd K, Vyletal P, Schaeffer C, Olinger E, Johnson E, Živná M, Jorge SC, Calado JT, Torres RJ, Lhotta K, Steubl D, Gale DP, Gast C, Gombos E, Robins V, Taylor A, Martin L, Ainsworth H, Chen Y, Almeida JR, Fernandes de Souza C, Silvera CS, Raposeiro R, Hodanova K, Votruba M, Vrbacká A, Divers J, Scolari F, Devuyst O, Rampoldi L, Kmoch S, Bleyer A. Genetic and clinical predictors of age of end-stage kidney disease in individuals with autosomal dominant tubulo-Interstitial kidney disease due to UMOD mutations. Kidney Int Rep 5(9): 1472-1485, 2020 PMCID: PMC7486199
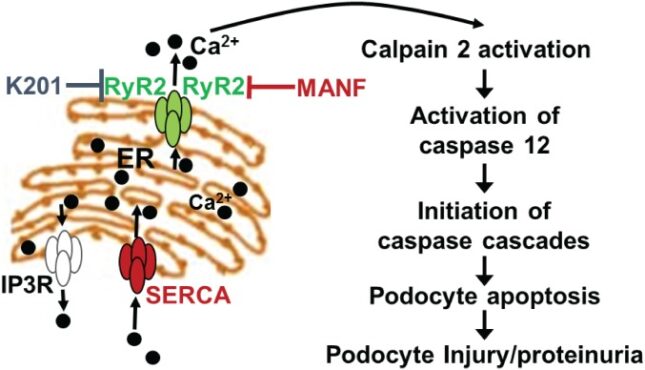
Park SJ, Kim Y, Yang SM, Henderson MJ, Yang W, Lindahl M, Urano F, Chen Y. Discovery of novel endoplasmic reticulum calcium stabilizers to rescue ER-stressed podocytes in nephrotic syndrome. Proc Natl Acad Sci 116(28): 14154-14163, 2019 PMCID: PMC6628787
Highlighted by Nature Reviews Nephrology in July 2019 https://www.nature.com/articles/s41581-019-0180-1
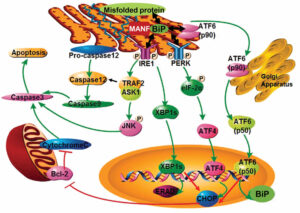
Park SJ, Kim Y, Chen Y. Endoplasmic reticulum stress and monogenic kidney diseases in precision nephrology. Pediatr Nephrol (https://doi.org/10.1007/s00467-018-4031-2), 2018 PMID: 30099615
Kim Y, Park SJ, Manson SR, Carlos M, Kidd K, Thiessen-Philbrook H, Perry R, Liapis H, Kmoch S, Parikh CR, Bleyer AJ, Chen Y. Elevated urinary CRELD2 is associated with endoplasmic reticulum stress-mediated kidney disease. JCI Insight 2(23): e92896 2017
Kim Y, Park SJ, Chen Y. Mesencephalic astrocyte-derived neurotrophic factor (MANF), a new player in endoplasmic reticulum diseases: structure, biology and therapeutic roles. Transl 188: 1–9 2017 [Featured New Investigator Series]
Mutneja A, Cossey LN, Liapis H, Chen Y. A rare case of renal thrombotic microangiopathy associated with Castleman’s disease. BMC Nephrol 18: 57, 2017.
Kim Y, Lee H, Manson SR, Lindahl M, Evans B, Miner JH, Urano F, Chen Y. Mesencephalic astrocyte-derived neurotrophic factor as a urine biomarker for endoplasmic reticulum stress-related kidney diseases. J Am Soc Nephrol 27: 2974-2982 2016 PMCID: PMC5042655
Singh S, Manson SR, Lee H, Kim Y, Liu T, Guo QS, Geminiani JJ, Austin PF, and Chen Y. Tubular overexpression of angiopoietin-1 attenuates renal fibrosis. PLOS ONE 11(7): e0158908 2016
Chen Y, Liapis H. Focal segmental glomerulosclerosis: molecular genetics and targeted therapies. [Corresponding author] BMC Nephrol 16: 101, 2015
Chen Y, Zhou YF, Go G, Marmerstein JT, Kikkawa Y, Miner J. Laminin β2 gene missense mutation produces endoplasmic reticulum stress in podocytes. J Am Soc Nephrol. 24: 1223-1233 2013
Chen Y, Marcos LA, Liapis H, Steinberg TH, Morrison AR. An unusual cause of membranous glomerulonephritis in a patient with HIV. Int Urol Nephrol. 44(3): 983-6 2012
Chen Y, Miner J. Glomerular basement membrane and related glomerular disease. Transl 160: 291–297 2012 [Corresponding author, Featured New Investigator Series]
Chen Y, Kikkawa Y, Miner J. A missense LAMB2 mutation causes congenital nephrotic syndrome by impairing laminin secretion. J Am Soc Nephrol. 22: 849-858 2011 [Cover illustration]
Chen Y, Donnelly E, Kobayashi H, DeBusk LM and Lin PC. Gene therapy targeting the Tie2 function ameliorates collagen-induced arthritis and protects against bone destruction. Arthritis Rheum. 52(5): 1585-1594 2005
[Editorial comments]: Pap T and Distler O. Linking angiogenesis to bone destruction in arthritis. Arthritis Rheum. 52(5): 1346-1348
Chen JX, Chen Y, DeBusk LM, Lin W, Lin PC. Dual functional roles of Tie2/angiopoietin in TNF-alpha-mediated angiogenesis. Am J Physiol Heart Circ Physiol. 287(1): H187-195 2004
DeBusk LM, Chen Y, Nishishita T, Chen J, Thomas JW, Lin PC. Tie2 receptor tyrosine kinase, a major mediator of tumor necrosis factor alpha-induced angiogenesis in rheumatoid arthritis. Arthritis Rheum. 48(9): 2461-2471 2003
Chen Y, Rosloniec E, Price J, Boothby M, Chen J. Constitutive expression of Bcl-xL in the T lineage attenuates collagen-induced arthritis in Bcl-xL transgenic mice. Arthritis Rheum. 46(2): 514-521 2002
Chen Y, Rosloniec E, Goral MI, Boothby M, Chen J. Redirection of T cell effector function in vivo and enhanced collagen-induced arthritis mediated by an IL-2Rb/IL-4Ra chimeric cytokine receptor transgene. Immunol. 166(6): 4163-4169 2001